Abstract
Background: CB transplantation (CBT) can be associated with the advantages of low rates of chronic GVHD & relapse. These benefits, however, can be offset by early post-transplant transplant-related mortality (TRM). Therefore, we have investigated CBT after a novel intermediate intensity, reduced toxicity myeloablative cyclophosphamide 50 mg/kg, fludarabine 150 mg/m2, thiotepa 5-10 mg/kg, 400 cGy TBI regimen in an effort to reduce early organ toxicity while also maximizing engraftment & disease control.
Methods: We analyzed CBT outcomes, including patient (pt) & graft characteristics associated with TRM, relapse and progression-free survival (PFS), in adult CBT recipients transplanted with this approach. Eligible pts were first allograft recipients </= 65 yrs transplanted for acute leukemia/ MDS/ MPD (</= 10% blasts pre-CBT), B-cell NHL or Hodgkin lymphoma. Graft-versus-host disease (GVHD) prophylaxis was with cyclosporine-A/ mycophenolate mofetil (no ATG).
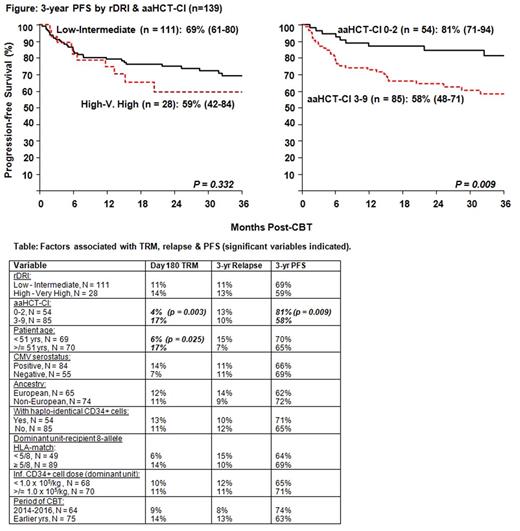
Results: 139 consecutive pts [74 (53%) non-European, 84 (60%) CMV seropositive, median age 51 yrs (range 23-65), median weight 83 kg (range 49-136)] were transplanted 10/2007-12/2016. Diagnoses included 102 acute leukemia, 22 MDS/ MPD, 15 B-cell NHL/ HL. All received double unit grafts [median donor recipient 8-allele HLA-match 5/8 (range 2-8), median infused CD34+ dose 1.0 x 105/kg/unit (range 0.2-8.6)]. 54 (39%) CB grafts were supplemented with haplo-identical CD34+ cells as a myeloid bridge. The incidence of CB engraftment was 96% (95%CI: 91-99). Day 180 incidence of grades II-IV & III-IV aGVHD was 78% (95%CI: 70-84) & 21% (95%CI: 15-28), respectively. 1-yr cGVHD was 8% (95%CI: 4-14). Cumulative incidence of day 180 TRM was 12% (95%CI: 7-17) & 21% (95%CI: 14-29) at 3 yrs. 11% (95%CI: 6-18) of pts relapsed by 3 yrs post-CBT. With a median survivor follow-up of 2.7 yrs (range 6.5 months-9.5 yrs), the 3-yr overall survival is 71% (95%CI: 63-80) & PFS is 67% (95%CI: 59-76). 3-yr PFS in 75 acute leukemia CR1 pts is 66% versus 74% in 27 other disease status pts (p = 0.574). 3-yr PFS in 23 FLT-3 mutated AML pts is 83%. The 3-yr PFS is 62% in 22 MDS/ MPD pts & 67% in 15 NHL/ HD pts. The revised disease risk index (rDRI) was not associated with TRM, relapse or PFS (Figure, Table). By contrast, pts with an age adjusted hematopoietic cell transplant-comorbidity index (aaHCT-CI) 0-2 (n = 54) had very low day 180 TRM [4% (95%CI: 1-11)] & high PFS [81% (95%CI: 71-94)] whereas the 85 pts with high aaHCT-CI (median 4, range 3-9) had a day 180 TRM of 17% (95%CI: 10-25) & 3-yr PFS of 58% (95%CI: 48-71). In multivariate analysis of TRM, aaHCT-CI had a HR of 4 (95%CI: 1.4-11.6, p = 0.01) whereas age alone was not significant [HR 1.9 (95%CI: 0.8-4.5), p = 0.12]. In multivariate analysis of PFS, aaHCT-CI HR was 2.4 (95%CI: 1.2-4.7, p = 0.01) whereas for recent pts (CBT 2014-2016) HR was 0.6 (95%CI: 0.3-1.2) although this was not significant (p = 0.17)
Conclusions: The 3-yr PFS in this older (median 51 yrs) intermediate intensity dCBT population is very promising. The incidences of cGVHD & relapse are low & DRI was not associated with TRM, relapse or PFS. Notably, addition of haplo-identical cells & better CB HLA-match was not associated with better PFS, & older age (51-65 yrs), CMV sero-positivity & non-European ancestry did not adversely affect PFS. Notably, day 180 TRM in recent pts is 9%. While high aaHCT-CI pts require additional strategies to reduce early post-CBT complications, given PFS is excellent in low aaHCT-CI CBT recipients (39% of pts in this analysis), survival comparisons of intermediate intensity dCBT in this population versus all other HSC sources is warranted. CB can also be attractive in pts with high risk disease given its rapid availability.
van den Brink: Seres: Research Funding; Therakos Institute: Other: Speaking engagement; PureTech Health: Consultancy; Jazz Pharmaceuticals: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal